Firstly, it is necessary to mention that in appearance purely theoretical aspects of corrosion (e.g. potential difference between anode and cathode, need for a complete electrical circuit, electrodes area, etc.) are of high importance in practice due to the professional involved will have an idea about what methods to take into account in order to avoid, as far as possible, the phenomenon of corrosion.
There are several ways in which corrosion occurs. The most relevant ones are indicated as follows, because of the importance that they have in the world of engineering.
- General corrosion. In this mechanism, the corrosion takes places in a relatively uniform way in exposed surface, so that a specially damaged zone cannot be considered to exist. It is usually a corrosion mechanism that happens on the surface of a metal where the anodes and the cathodes are caused by little differences between the different zones of corroded surface.
- Pitting corrosion. It is a type of localized corrosion in which a punctual area of material is attacked but a great penetration there happens. In this kind of corrosion, the speed at which the perforation in the metal occurs is quite high. That is why pitting corrosion is considered as very harmful.
- Crevice corrosion. Another type of localized corrosion but now the attack area has a restricted access to environment. This type of corrosion is caused by a difference in the materials concentration between interior and exterior zones of the crevice.
- Galvanic corrosion. This way takes place due to potential difference (i.e. energy level) between metal in electric contact.
- Intergranular corrosion. This type occurs preferably in the grain boundaries of a metal.
- Fatigue corrosion. It is caused by a combination of a variable stress (fatigue) and a corrosion environment.
It should be noted that, in most of the cases that will be focused, metal existing in the same industrial facility will be subjected to a combination to several corrosion ways, so the solution applied must consider all harmful effects.
Some of the most commonly used methods to avoid the main ways of corrosion are exposed as follows.
- When the engineer faces a general corrosion case in which the corrosion speed is relatively low, especially if speed decreased in function of time, this kind of corrosion could be assumed without establishing a method to avoid it. During stage of design of component that will be subjected to general corrosion, a specialist could calculate the thickness losses that will happen in order to add an additional thickness that is named as corrosion allowance.
- As it is well-known, not all the metals and their alloys are equally resistant to corrosion effects. For this reason, a quite usual solution is to use alloys named stainless steel. We have to keep an eye on an economic factor, that the alloying components that exist in stainless steel increase the cost of the metal, and a technical factor, as each type of stainless steel has an effective resistance against some specific kind of corrosion.
- Changes that we could implement in design of components must always be taken into account as they have a lower cost impact and they are easier to implement than in later stage of manufacturing or operation of the equipment. This could involve subjects as a change in geometry of the element in order to avoid zones where the water can be retained, as to avoid two material with different electric potential in direct contact to elude galvanic corrosion, and as significant variations in operating conditions which affects to component.
- Another way that it is convenient to keep in mind is the modification of the environment. Regarding this concept, we could find the corrosion inhibitors, which consist of substances that, if they are added to environment, decrease the corrosion attack speed because of existing environment. They generally attenuate corrosion due to generate thin films that modify the environment that affects the metal.
- Protective coatings are especially effective in corrosion control and probably they are the most commonly used method. Protective coating are something so, in appearance, basic as to interpose a separating barrier between the metal to be protected against corrosion and the environment that tends to corrode it.
- Cathodic protection is a way to avoid corrosion that interferes with current flow in the electrochemical cell and, among other situations, is used in equipment that are buried or immersed. This protective mechanism has a wide range of environments in which its use is effective. It reduces, or removes, corrosion by means of causing the material to be protected is converted in a cathode using an impressed current or adding an anode, commonly materials as zinc or magnesium.
- Unlike cathodic protection, anodic protection has a much smaller scope, especially dedicated to chemical environments. It works by means of maintaining an active-passive metal within the passive zone. For that, an anodic current is applied externally, as it is graphically shown as follows.
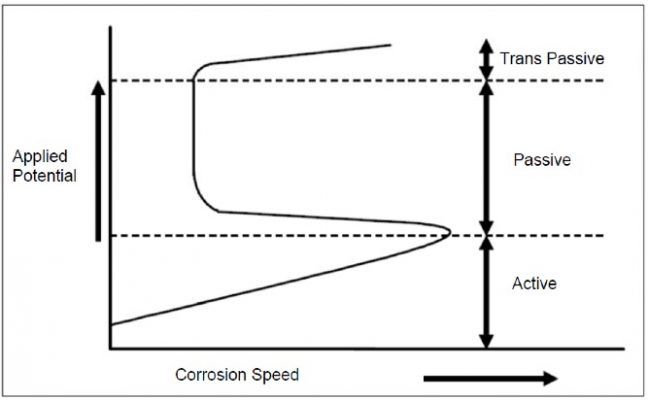
Passivation could be achieved by modifying the metal potential by means of an external source.
If you want to know more:
ASME VIII | Design of Pressure Vessels
TEMA | Design of Shell & Tube Heat Exchangers


 WhatsApp
WhatsApp