Material selection for special services

In the processes that are carried out, both in refineries and in chemical plants, to obtain different products, we can verify the formation of a multitude of reactions that are necessary to achieve the proposed objectives.
The main objective of these process reactions is to obtain a final product free of all types of contaminants and impurities contained in the raw materials and thus comply with the requirements of the corresponding specifications. To this end, the equipment in which such processes are carried out must be constructed of Carbon Steel materials but subject to certain restrictions, limitations and particular conditions required to withstand the failure mechanisms derived from the presence of different corrosive agents and/or operating conditions such as low and high temperature.
In the API RP571 standard “Damage Mechanisms Affecting Fixed Equipment in the Refining Industry” we can find a list with the different degradation mechanisms that can be seen in the different process units of a refinery.
Low Temperature:
When the low temperature is mentioned, it’s associated with the embrittlement shown in many Carbon and Low Alloy Steel materials .
Perhaps it is one of the issues that most concern generated to ASME and other design codes from the beginning.. The reason is the loss of toughness and decrease in impact properties experienced by these materials at a temperature below the ductile-brittle transition one.
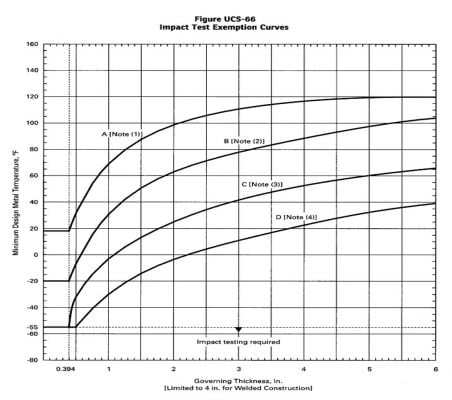
Impact Test Exception Curves
Services with wet hydrogen sulfide:
The hydrogen sulfide is one of the most harmful contaminants and depending on H2S content, atomic H coming from the dissociation of H2S absorbed and the pH level and temperature, results in the development of one of the following degradation mechanisms in Carbon Steel:
- SSCC (Sulfide Stress Corrosion Cracking)
- HIC (Hydrogen Induced Cracking)
- SOHIC (Strees Oriented Hydrogen Induced Cracking)
- Blistering
Measurements to avoid or minimize the above effects:
- Chemical composition restriction
- Steel cleaning: extremely low residual elements content.
- Inclusions control
- Post Weld Heat Treatment
- Hardness Control
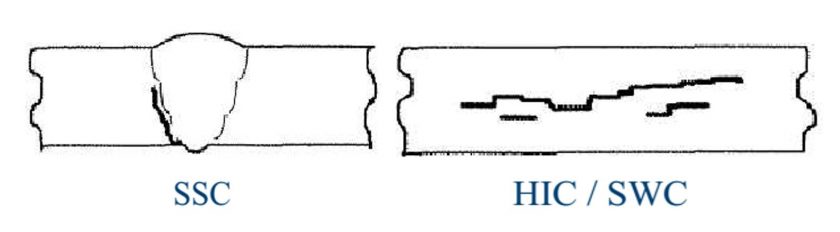
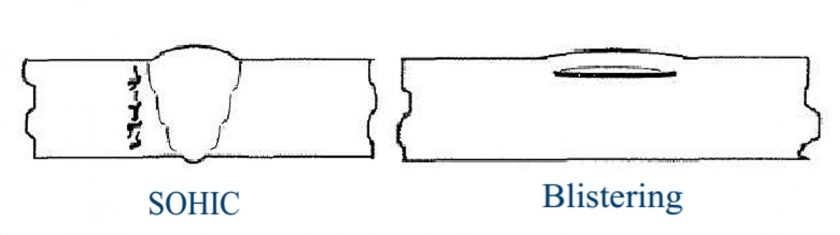
Caustic Service:
The presence of Caustic Soda (NaOH) causes the material to become susceptible to embrittlement and consequently to cracking.
Soda attacks both carbon Steel; series 300 Stainless Steel, and to a lesser extent Ni-based alloys. Ni alloys are highly resistant (although not immune).
The aggressiveness is a function of NaOH content, temperature, and the material residual stress level.
The NACE SP0403 standard is used as a reference guide for selecting the most appropriate material of construction for equipment handling soda solution, based on NaOH content and temperature.
The most effective method to prevent this phenomenon in the case of Carbon Steel, is through the application of PWHT.
Temper Embrittlement:
This is the loss of the material’s toughness properties, which although occurring in a temperature range of 350-600°C, only manifests itself during the shutdown and/or start-up stages. Embrittled material can then led to brittle fracture.
The presence of tramp elements such as Phosphorus, Arsenic, Antimony and Tin is extremely dangerous and must be carefully controlled.
The so-called Step Cooling Test is the preferred method to predict whether the material becomes embrittled during exposure to operating temperature, by impact testing of specimens subjected to this treatment.
Through this test, we can determine if there is a shift in the ductile-brittle transition curve of the material subjected to SC compared with the original curve.
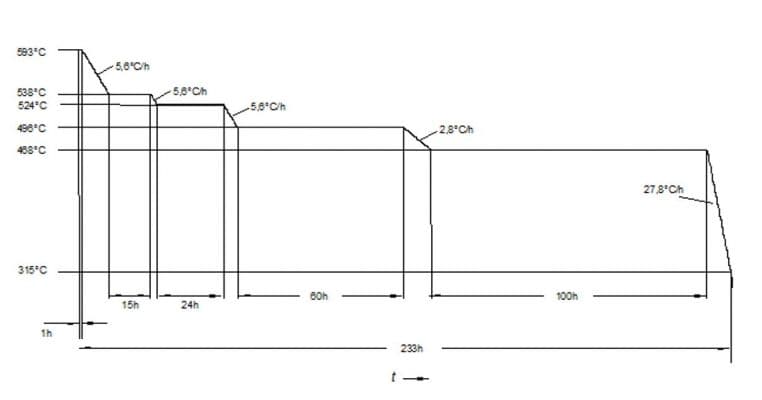
Step Cooling Procedure
High Temperature Hydrogen Attack:
It occurs when materials are exposed to a temperature greater than 204º C in a Hydrogen-containing environment, depending on the Hydrogen partial pressure, temperature, and time exposure.
Causes: Atomic hydrogen permeating the steel reacts with C to form CH4 (methane) molecules. This formed methane gas cannot diffuse and led to the formation of fissures, cracks, and blistering in the steel.
On the other hand, a decrease in the mechanical properties can also be observed, due to the decarburization process that takes place simultaneously.
The most common practice to increase the resistance against H attack to CS is the application of PWHT. Also, the use of low alloy steel with Cr and Mo, depending on the operating temperature of the equipment.
The API RP941 standard includes Nelson curves, a guide for the selection of the most suitable material as a function of hydrogen partial pressure, and the operating temperature.
If you want to know more:

 WhatsApp
WhatsApp